Fleur M. Ferguson, Zainab M. Doctor, Scott B. Ficarro, Jarrod A. Marto, Nam Doo Kim, Taebo Sim, Nathanael S. Gray
Bioorganic & Medicinal Chemistry Letters, 2019
doi: 10.1016/j.bmcl.2019.05.024
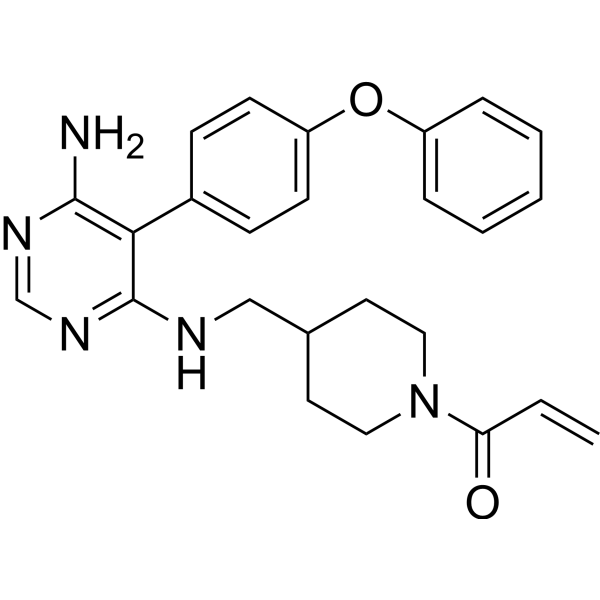
The TAIRE family of kinases are an understudied branch of the CDK kinase family, that have been implicated in a number of cancers. This manuscript describes the design, synthesis and SAR of covalent CDK14 inhibitors, culminating in identification of FMF-04-159-2, a potent, covalent CDK14 inhibitor with a TAIRE kinase biased selectivity profile.
A blog highlighting recent publications in the area of covalent modification of proteins, particularly relating to covalent-modifier drugs. @CovalentMod on Twitter, @covalentmod@mstdn.science on Mastodon, and @covalentmod.bsky.social on BlueSky
Tuesday, May 28, 2019
Thursday, May 23, 2019
Traceless Templated Amide-Forming Ligations [@bode_lab]
Alberto Osuna Gálvez, Jeffrey W. Bode
J. Am. Chem. Soc. 2019
DOI: 10.1021/jacs.9b03597
Template assistance allows organic reactions to occur under highly diluted conditions – where intermolecular reactions often fail to proceed – by bringing reactants into close spatial proximity. This strategy has been elegantly applied to numerous systems, but always with the retention of at least one of the templating groups in the product. In this report, we describe a traceless, templated amide-forming ligation that proceeds at low micromolar concentration under aqueous conditions in the presence of biomolecules. We utilized the unique features of an acylboronate–hydroxylamine ligation, in which covalent bonds are broken in each of the reactants as the new amide bond is formed. By using streptavidin as a template and acylboronates and O–acylhydroxylamines bearing desthiobiotins that are cleaved upon amide formation, we demonstrate that traceless, templated ligation occurs rapidly even at sub-micromolar concentrations. The requirement for a close spatial orientation of the functional groups – achieved upon binding to streptavidin – is critical for the observed enhancement in the rate and quantity of product formed.
DOI: 10.1021/jacs.9b03597
Template assistance allows organic reactions to occur under highly diluted conditions – where intermolecular reactions often fail to proceed – by bringing reactants into close spatial proximity. This strategy has been elegantly applied to numerous systems, but always with the retention of at least one of the templating groups in the product. In this report, we describe a traceless, templated amide-forming ligation that proceeds at low micromolar concentration under aqueous conditions in the presence of biomolecules. We utilized the unique features of an acylboronate–hydroxylamine ligation, in which covalent bonds are broken in each of the reactants as the new amide bond is formed. By using streptavidin as a template and acylboronates and O–acylhydroxylamines bearing desthiobiotins that are cleaved upon amide formation, we demonstrate that traceless, templated ligation occurs rapidly even at sub-micromolar concentrations. The requirement for a close spatial orientation of the functional groups – achieved upon binding to streptavidin – is critical for the observed enhancement in the rate and quantity of product formed.
Sunday, May 19, 2019
Development of a Covalent Inhibitor of Gut Bacterial Bile Salt Hydrolases [@DevlinLab]
Arijit A. Adhikari, Tom C. Seegar, Scott B. Ficarro, Megan D. McCurry, Deepti Ramachandran, Lina Yao, Snehal N. Chaudhari, Sula Ndousse-Fetter, Alexander S. Banks, Jarrod A. Marto, Stephen C. Blacklow, A. Sloan Devlin
BioRxiv, 2019
doi: 10.1101/640086
Bile salt hydrolase (BSH) enzymes are widely expressed by human gut bacteria and catalyze the gateway reaction leading to secondary bile acid formation. Bile acids regulate key metabolic and immune processes by binding to host receptors. There is an unmet need for a potent tool to inhibit BSHs across all gut bacteria in order to study the effects of bile acids on host physiology. Here, we report the development of a covalent pan-inhibitor of gut bacterial BSH. From a rationally designed candidate library, we identified a lead compound bearing an alpha-fluoromethyl ketone warhead that modifies BSH at the catalytic cysteine residue. Strikingly, this inhibitor abolished BSH activity in conventional mouse feces. Mice gavaged with a single dose of this compound displayed decreased BSH activity and decreased deconjugated bile acid levels in feces. Our studies demonstrate the potential of a covalent BSH inhibitor to modulate bile acid composition in vivo.
BioRxiv, 2019
doi: 10.1101/640086
Bile salt hydrolase (BSH) enzymes are widely expressed by human gut bacteria and catalyze the gateway reaction leading to secondary bile acid formation. Bile acids regulate key metabolic and immune processes by binding to host receptors. There is an unmet need for a potent tool to inhibit BSHs across all gut bacteria in order to study the effects of bile acids on host physiology. Here, we report the development of a covalent pan-inhibitor of gut bacterial BSH. From a rationally designed candidate library, we identified a lead compound bearing an alpha-fluoromethyl ketone warhead that modifies BSH at the catalytic cysteine residue. Strikingly, this inhibitor abolished BSH activity in conventional mouse feces. Mice gavaged with a single dose of this compound displayed decreased BSH activity and decreased deconjugated bile acid levels in feces. Our studies demonstrate the potential of a covalent BSH inhibitor to modulate bile acid composition in vivo.
Thursday, May 16, 2019
Covalent Ligand Screening Uncovers a RNF4 E3 Ligase Recruiter for Targeted Protein Degradation Applications [@DanNomura]
Carl C. Ward, Jordan I. Kleinman, Scott M. Brittain, Patrick S. Lee, Clive Yik Sham Chung, Kenneth Kim, Yana Petri, Jason R. Thomas, John A. Tallarico, Jeffrey M. McKenna, Markus Schirle, and Daniel K. Nomura
ACS Chemical Biology 2019
DOI: 10.1021/acschembio.8b01083Targeted protein degradation has arisen as a powerful strategy for drug discovery allowing the targeting of undruggable proteins for proteasomal degradation. This approach most often employs heterobifunctional degraders consisting of a protein-targeting ligand linked to an E3 ligase recruiter to ubiquitinate and mark proteins of interest for proteasomal degradation. One challenge with this approach, however, is that only a few E3 ligase recruiters currently exist for targeted protein degradation applications, despite the hundreds of known E3 ligases in the human genome. Here, we utilized activity-based protein profiling (ABPP)-based covalent ligand screening approaches to identify cysteine-reactive small-molecules that react with the E3 ubiquitin ligase RNF4 and provide chemical starting points for the design of RNF4-based degraders. The hit covalent ligand from this screen reacted with either of two zinc-coordinating cysteines in the RING domain, C132 and C135, with no effect on RNF4 activity. We further optimized the potency of this hit and incorporated this potential RNF4 recruiter into a bifunctional degrader linked to JQ1, an inhibitor of the BET family of bromodomain proteins. We demonstrate that the resulting compound CCW 28-3 is capable of degrading BRD4 in a proteasome- and RNF4-dependent manner. In this study, we have shown the feasibility of using chemoproteomics-enabled covalent ligand screening platforms to expand the scope of E3 ligase recruiters that can be exploited for targeted protein degradation applications.
Sunday, May 12, 2019
Placebo-Controlled Trial of an Oral BTK Inhibitor in Multiple Sclerosis
Xavier Montalban, M.D., Ph.D., Douglas L. Arnold, M.D., Martin S. Weber, M.D., Ivan Staikov, M.D., Ph.D., Karolina Piasecka-Stryczynska, M.D., Ph.D., Jonathan Willmer, M.D., Emily C. Martin, Ph.D., Fernando Dangond, M.D., Sana Syed, M.D., M.P.H., and Jerry S. Wolinsky, M.D.
New England Journal of Medicine, 2019
doi: 10.1056/nejmoa1901981
Bruton’s tyrosine kinase (BTK) regulates the functions of B cells and myeloid cells that are implicated in the pathogenesis of multiple sclerosis. Evobrutinib is a selective oral BTK inhibitor that has been shown to inhibit B-cell activation both in vitro and in vivo.
METHODS
In this double-blind, randomized, phase 2 trial, we assigned patients with relapsing multiple sclerosis to one of five groups: placebo, evobrutinib (at a dose of 25 mg once daily, 75 mg once daily, or 75 mg twice daily), or open-label dimethyl fumarate (DMF) as a reference. The primary end point was the total (cumulative) number of gadolinium-enhancing lesions identified on T1-weighted magnetic resonance imaging at weeks 12, 16, 20, and 24. Key secondary end points included the annualized relapse rate and change from baseline in the score on the Expanded Disability Status Scale (EDSS).
RESULTS
A total of 267 patients were randomly assigned to a trial group. The mean (±SD) total number of gadolinium-enhancing lesions during weeks 12 through 24 was 3.85±5.44 in the placebo group, 4.06±8.02 in the evobrutinib 25-mg group, 1.69±4.69 in the evobrutinib 75-mg once-daily group, 1.15±3.70 in the evobrutinib 75-mg twice-daily group, and 4.78±22.05 in the DMF group. The baseline adjusted rate ratios for the total number of lesions over time as compared with placebo were 1.45 in the evobrutinib 25-mg group (P=0.32), 0.30 in the evobrutinib 75-mg once-daily group (P=0.005), and 0.44 in the evobrutinib 75-mg twice-daily group (P=0.06). The unadjusted annualized relapse rate at week 24 was 0.37 in the placebo group, 0.57 in the evobrutinib 25-mg group, 0.13 in the evobrutinib 75-mg once-daily group, 0.08 in the evobrutinib 75-mg twice-daily group, and 0.20 in the DMF group. There was no significant effect of trial group on the change from baseline in the EDSS score. Elevations in liver aminotransferase values were observed with evobrutinib.
CONCLUSIONS
Patients with relapsing multiple sclerosis who received 75 mg of evobrutinib once daily had significantly fewer enhancing lesions during weeks 12 through 24 than those who received placebo. There was no significant difference with placebo for either the 25-mg once-daily or 75-mg twice-daily dose of evobrutinib, nor in the annualized relapse rate or disability progression at any dose. Longer and larger trials are required to determine the effect and risks of evobrutinib in patients with multiple sclerosis. (Funded by EMD Serono; ClinicalTrials.gov number, NCT02975349.)
New England Journal of Medicine, 2019
doi: 10.1056/nejmoa1901981
Bruton’s tyrosine kinase (BTK) regulates the functions of B cells and myeloid cells that are implicated in the pathogenesis of multiple sclerosis. Evobrutinib is a selective oral BTK inhibitor that has been shown to inhibit B-cell activation both in vitro and in vivo.
METHODS
In this double-blind, randomized, phase 2 trial, we assigned patients with relapsing multiple sclerosis to one of five groups: placebo, evobrutinib (at a dose of 25 mg once daily, 75 mg once daily, or 75 mg twice daily), or open-label dimethyl fumarate (DMF) as a reference. The primary end point was the total (cumulative) number of gadolinium-enhancing lesions identified on T1-weighted magnetic resonance imaging at weeks 12, 16, 20, and 24. Key secondary end points included the annualized relapse rate and change from baseline in the score on the Expanded Disability Status Scale (EDSS).
RESULTS
A total of 267 patients were randomly assigned to a trial group. The mean (±SD) total number of gadolinium-enhancing lesions during weeks 12 through 24 was 3.85±5.44 in the placebo group, 4.06±8.02 in the evobrutinib 25-mg group, 1.69±4.69 in the evobrutinib 75-mg once-daily group, 1.15±3.70 in the evobrutinib 75-mg twice-daily group, and 4.78±22.05 in the DMF group. The baseline adjusted rate ratios for the total number of lesions over time as compared with placebo were 1.45 in the evobrutinib 25-mg group (P=0.32), 0.30 in the evobrutinib 75-mg once-daily group (P=0.005), and 0.44 in the evobrutinib 75-mg twice-daily group (P=0.06). The unadjusted annualized relapse rate at week 24 was 0.37 in the placebo group, 0.57 in the evobrutinib 25-mg group, 0.13 in the evobrutinib 75-mg once-daily group, 0.08 in the evobrutinib 75-mg twice-daily group, and 0.20 in the DMF group. There was no significant effect of trial group on the change from baseline in the EDSS score. Elevations in liver aminotransferase values were observed with evobrutinib.
CONCLUSIONS
Patients with relapsing multiple sclerosis who received 75 mg of evobrutinib once daily had significantly fewer enhancing lesions during weeks 12 through 24 than those who received placebo. There was no significant difference with placebo for either the 25-mg once-daily or 75-mg twice-daily dose of evobrutinib, nor in the annualized relapse rate or disability progression at any dose. Longer and larger trials are required to determine the effect and risks of evobrutinib in patients with multiple sclerosis. (Funded by EMD Serono; ClinicalTrials.gov number, NCT02975349.)

Tuesday, May 7, 2019
Rapid covalent-probe discovery by electrophile-fragment screening [@london_lab]
Efrat Resnick, Anthony Bradley, Jinrui Gan, Alice Douangamath, Tobias Krojer, Ritika Sethi, Paul P. Geurink, Anthony Aimon, Gabriel Amitai, Dom Bellini, James Bennett, Michael Fairhead, Oleg Fedorov, Ronen Gabizon, jin gan, Jingxu Guo, Alexander Plotnikov, Nava Reznik, Gian Filippo Ruda, Laura Díaz-Sáez, Verena M. Straub, Tamas Szommer, Srikannathasan Velupillai, Daniel Zaidman, Yanling Zhang, Alun R. Coker, Christopher G Dowson, Haim Barr, Chu Wang, Kilian V. M. Huber, Paul E Brennan, Huib Ovaa, Frank von Delft, and Nir London
Journal of the American Chemical Society 2019
DOI: 10.1021/jacs.9b02822Covalent probes can display unmatched potency, selectivity and duration of action; however, their discovery is challenging. In principle, fragments that can irreversibly bind their target can overcome the low affinity that limits reversible fragment screening, but such electrophilic fragments were considered non-selective and were rarely screened. We hypothesized that mild electrophiles might overcome the selectivity challenge and constructed a library of 993 mildly electrophilic fragments. We characterized this library by a new high-throughput thiol-reactivity assay and screened them against ten cysteine-containing proteins. Highly reactive and promiscuous fragments were rare and could be easily eliminated. By contrast, we found hits for most targets. Combining our approach with high-throughput crystallography allowed rapid progression to potent and selective probes for two enzymes, the deubiquitinase OTUB2 and the pyrophosphatase NUDT7. No inhibitors were previously known for either. This study highlights the potential of electrophile-fragment screening as a practical and efficient tool for covalent-ligand discovery.
Subscribe to:
Posts (Atom)
A chemoproteomic atlas of the human purine interactome for regioselective ligand discovery
Zhihong Li, Hsiao-Kuei Tsai, Adam H. Libby, Michael W. Founds, Olivia L. Murtagh, Madeleine L. Ware, David M. Leace, Wesley J. Wolfe, Philli...
-
Design, synthesis and biological evaluation of the activity-based probes for FGFR covalent inhibitorDandan Zhu, Zijian Zheng, Huixin Huang, Xiaojuan Chen, Shuhong Zhang, Zhuchu Chen, Ting Liu, Guangyu Xu, Ying Fu, Yongheng Chen, European Jo...
-
Yoav Shamir, Nir London bioRxiv 2025.03.19.642201 doi: https://doi.org/10.1101/2025.03.19.642201 Recent years have seen an explosion in the...
-
DOI Ansgar Oberheide, Maxime van den Oetelaar, Jakob Scheele, Jan Borggräfe, Semmy Engelen, Michael Sattler, Christian Ottmann, ...




