John K. Eaton, Laura Furst, Richard A. Ruberto, Dieter Moosmayer, André Hilpmann, Matthew J. Ryan, Katja Zimmermann, Luke L. Cai, Michael Niehues, Volker Badock, Anneke Kramm, Sixun Chen, Roman C. Hillig, Paul A. Clemons, Stefan Gradl, Claire Montagnon, Kiel E. Lazarski, Sven Christian, Besnik Bajrami, Roland Neuhaus, Ashley L. Eheim, Vasanthi S. Viswanathan & Stuart L. Schreiber
Nature Chemical Biology, 2020
DOI: https://doi.org/10.1038/s41589-020-0501-5
We recently described glutathione peroxidase 4 (GPX4) as a promising target for killing therapy-resistant cancer cells via ferroptosis. The onset of therapy resistance by multiple types of treatment results in a stable cell state marked by high levels of polyunsaturated lipids and an acquired dependency on GPX4. Unfortunately, all existing inhibitors of GPX4 act covalently via a reactive alkyl chloride moiety that confers poor selectivity and pharmacokinetic properties. Here, we report our discovery that masked nitrile-oxide electrophiles, which have not been explored previously as covalent cellular probes, undergo remarkable chemical transformations in cells and provide an effective strategy for selective targeting of GPX4. The new GPX4-inhibiting compounds we describe exhibit unexpected proteome-wide selectivity and, in some instances, vastly improved physiochemical and pharmacokinetic properties compared to existing chloroacetamide-based GPX4 inhibitors. These features make them superior tool compounds for biological interrogation of ferroptosis and constitute starting points for development of improved inhibitors of GPX4.
A blog highlighting recent publications in the area of covalent modification of proteins, particularly relating to covalent-modifier drugs. @CovalentMod on Twitter, @covalentmod@mstdn.science on Mastodon, and @covalentmod.bsky.social on BlueSky
Monday, March 30, 2020
Saturday, March 28, 2020
Insight Into the Therapeutic Selectivity of the Irreversible EGFR Tyrosine Kinase Inhibitor Osimertinib Through Enzyme Kinetic Studies
Xiang Zhai, Richard A Ward, Peter Doig, and Argyrides Argyrou
Biochemistry 2020
DOI: 10.1021/acs.biochem.0c00104
Osimertinib is a covalent, third-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) approved for treating non-small cell lung cancer (NSCLC) patients with activating EGFR mutations (Exon19del or L858R), or with T790M resistance mutation following disease progression on first- or second-generation EGFR TKIs. The aim of this work was to understand how osimertinib achieves selective inhibition of mutant EGFR relative to wildtype through evaluating its kinetic mechanism of action. In doing so, we developed methodologies combining steady-state and pre-steady-state kinetics to determine the covalent inactivation rates (kinact) and reversible binding affinities (Ki) of osimertinib for WT, L858R and L858R/T790M EGFR, and compared these data to the inhibition kinetics of earlier generations of EGFR TKIs. The kinact/KI values indicate that osimertinib inactivates L858R and L858R/T790M with 20-fold and 50-fold higher overall efficiencies compared to WT, respectively. The Ki values reveal that osimertinib binds 3-fold and 17-fold tighter to L858R and L858R/T790M than to WT, respectively, while the kinact values show that osimertinib reacts 3-fold faster with L858R and L858R/T790M than with WT EGFR. We conclude that osimertinib achieves selective inhibition of L858R and L858R/T790M through improved affinities and improved rates of covalent bond formation via better positioning of the acrylamide warhead. This work highlights the importance of optimizing both reversible drug-target interactions and the inactivation rates for covalent inhibitors to achieve selectivity targeting mutant EGFR.
Biochemistry 2020
DOI: 10.1021/acs.biochem.0c00104
Osimertinib is a covalent, third-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) approved for treating non-small cell lung cancer (NSCLC) patients with activating EGFR mutations (Exon19del or L858R), or with T790M resistance mutation following disease progression on first- or second-generation EGFR TKIs. The aim of this work was to understand how osimertinib achieves selective inhibition of mutant EGFR relative to wildtype through evaluating its kinetic mechanism of action. In doing so, we developed methodologies combining steady-state and pre-steady-state kinetics to determine the covalent inactivation rates (kinact) and reversible binding affinities (Ki) of osimertinib for WT, L858R and L858R/T790M EGFR, and compared these data to the inhibition kinetics of earlier generations of EGFR TKIs. The kinact/KI values indicate that osimertinib inactivates L858R and L858R/T790M with 20-fold and 50-fold higher overall efficiencies compared to WT, respectively. The Ki values reveal that osimertinib binds 3-fold and 17-fold tighter to L858R and L858R/T790M than to WT, respectively, while the kinact values show that osimertinib reacts 3-fold faster with L858R and L858R/T790M than with WT EGFR. We conclude that osimertinib achieves selective inhibition of L858R and L858R/T790M through improved affinities and improved rates of covalent bond formation via better positioning of the acrylamide warhead. This work highlights the importance of optimizing both reversible drug-target interactions and the inactivation rates for covalent inhibitors to achieve selectivity targeting mutant EGFR.
Thursday, March 26, 2020
Sulfopin, a selective covalent inhibitor of Pin1, blocks Myc-driven tumor initiation and growth in vivo
Christian Dubiella, Benika J. Pinch, Daniel Zaidman, Theresa D. Manz, Evon Poon, Shuning He, Efrat Resnick, Ellen M. Langer, Colin J. Daniel, Hyuk-Soo Seo, Ying Chen, Scott B. Ficarro, Yann Jamin, Xiaolan Lian, Shin Kibe, Shingo Kozono, Kazuhiro Koikawa, Zainab M. Doctor, Behnam Nabet, Christopher M. Browne, Annan Yang, Liat Stoler-Barak, Richa B. Shah, Nick E. Vangos, Ezekiel A. Geffken, Roni Oren, Samuel Sidi, Ziv Shulman, Chu Wang, Jarrod A. Marto, Sirano Dhe-Paganon, Thomas Look, Xiao Zhen Zhou, Kun Ping Lu, Rosalie C. Sears, Louis Chesler, Nathanael S. Gray, Nir London
BioRXiv, 2020
doi: https://doi.org/10.1101/2020.03.20.998443
The peptidyl-prolyl cis-trans isomerase, Pin1, acts as a unified signaling hub that is exploited in cancer to activate oncogenes and inactivate tumor suppressors, in particular through up-regulation of c-Myc target genes. However, despite considerable efforts, Pin1 has remained an elusive drug target. Here, we screened an electrophilic fragment library to discover covalent inhibitors targeting Pin1’s active site nucleophile - Cys113, leading to the development of Sulfopin, a double-digit nanomolar Pin1 inhibitor. Sulfopin is highly selective for Pin1, as validated by two independent chemoproteomics methods, achieves potent cellular and in vivo target engagement, and phenocopies genetic knockout of Pin1. Although Pin1 inhibition had a modest effect on viability in cancer cell cultures, Sulfopin induced downregulation of c-Myc target genes and reduced tumor initiation and tumor progression in murine and zebrafish models of MYCN-driven neuroblastoma. Our results suggest that Sulfopin is a suitable chemical probe for assessing Pin1-dependent pharmacology in cells and in vivo. Moreover, these studies indicate that Pin1 should be further investigated as a potential cancer target.
BioRXiv, 2020
doi: https://doi.org/10.1101/2020.03.20.998443
The peptidyl-prolyl cis-trans isomerase, Pin1, acts as a unified signaling hub that is exploited in cancer to activate oncogenes and inactivate tumor suppressors, in particular through up-regulation of c-Myc target genes. However, despite considerable efforts, Pin1 has remained an elusive drug target. Here, we screened an electrophilic fragment library to discover covalent inhibitors targeting Pin1’s active site nucleophile - Cys113, leading to the development of Sulfopin, a double-digit nanomolar Pin1 inhibitor. Sulfopin is highly selective for Pin1, as validated by two independent chemoproteomics methods, achieves potent cellular and in vivo target engagement, and phenocopies genetic knockout of Pin1. Although Pin1 inhibition had a modest effect on viability in cancer cell cultures, Sulfopin induced downregulation of c-Myc target genes and reduced tumor initiation and tumor progression in murine and zebrafish models of MYCN-driven neuroblastoma. Our results suggest that Sulfopin is a suitable chemical probe for assessing Pin1-dependent pharmacology in cells and in vivo. Moreover, these studies indicate that Pin1 should be further investigated as a potential cancer target.
Friday, March 20, 2020
Crystal structure of SARS-CoV-2 main protease provides a basis for design of improved α-ketoamide inhibitors
Linlin Zhang, Daizong Lin, Xinyuanyuan Sun, Ute Curth, Christian Drosten, Lucie Sauerhering, Stephan Becker, Katharina Rox, Rolf Hilgenfeld
Science, 2020: eabb3405
DOI: 10.1126/science.abb3405
The COVID-19 pandemic caused by SARS-CoV-2 is a global health emergency. An attractive drug target among coronaviruses is the main protease (Mpro, 3CLpro), due to its essential role in processing the polyproteins that are translated from the viral RNA. We report the X-ray structures of the unliganded SARS-CoV-2 Mpro and its complex with an α-ketoamide inhibitor. This was derived from a previously designed inhibitor but with the P3-P2 amide bond incorporated into a pyridone ring to enhance the half-life of the compound in plasma. Based on the structure, we developed the lead compound into a potent inhibitor of the SARS-CoV-2 Mpro. The pharmacokinetic characterization of the optimized inhibitor reveals a pronounced lung tropism and suitability for administration by the inhalative route.
Science, 2020: eabb3405
DOI: 10.1126/science.abb3405
The COVID-19 pandemic caused by SARS-CoV-2 is a global health emergency. An attractive drug target among coronaviruses is the main protease (Mpro, 3CLpro), due to its essential role in processing the polyproteins that are translated from the viral RNA. We report the X-ray structures of the unliganded SARS-CoV-2 Mpro and its complex with an α-ketoamide inhibitor. This was derived from a previously designed inhibitor but with the P3-P2 amide bond incorporated into a pyridone ring to enhance the half-life of the compound in plasma. Based on the structure, we developed the lead compound into a potent inhibitor of the SARS-CoV-2 Mpro. The pharmacokinetic characterization of the optimized inhibitor reveals a pronounced lung tropism and suitability for administration by the inhalative route.
Thursday, March 19, 2020
Small-molecule covalent bond formation at tyrosine creates a binding site and inhibits activation of Ral GTPases
Khuchtumur Bum-Erdene, Degang Liu, Giovanni Gonzalez-Gutierrez, Mona K. Ghozayel, David Xu, Samy O. Meroueh
Proceedings of the National Academy of Science, 2020, 201913654;
DOI: 10.1073/pnas.1913654117
Ral (Ras-like) GTPases are directly activated by oncogenic Ras GTPases. Mutant K-Ras (G12C) has enabled the development of covalent K-Ras inhibitors currently in clinical trials. However, Ral, and the overwhelming majority of mutant oncogenic K-Ras, are devoid of a druggable pocket and lack an accessible cysteine for the development of a covalent inhibitor. Here, we report that covalent bond formation by an aryl sulfonyl fluoride electrophile at a tyrosine residue (Tyr-82) inhibits guanine exchange factor Rgl2-mediated nucleotide exchange of Ral GTPase. A high-resolution 1.18-Å X-ray cocrystal structure shows that the compound binds to a well-defined binding site in RalA as a result of a switch II loop conformational change. The structure, along with additional high-resolution crystal structures of several analogs in complex with RalA, confirm the importance of key hydrogen bond anchors between compound sulfone oxygen atoms and Ral backbone nitrogen atoms. Our discovery of a pocket with features found on known druggable sites and covalent modification of a bystander tyrosine residue present in Ral and Ras GTPases provide a strategy that could lead to therapeutic agent targeting oncogenic Ras mutants that are devoid of a cysteine nucleophile.
Proceedings of the National Academy of Science, 2020, 201913654;
DOI: 10.1073/pnas.1913654117
Ral (Ras-like) GTPases are directly activated by oncogenic Ras GTPases. Mutant K-Ras (G12C) has enabled the development of covalent K-Ras inhibitors currently in clinical trials. However, Ral, and the overwhelming majority of mutant oncogenic K-Ras, are devoid of a druggable pocket and lack an accessible cysteine for the development of a covalent inhibitor. Here, we report that covalent bond formation by an aryl sulfonyl fluoride electrophile at a tyrosine residue (Tyr-82) inhibits guanine exchange factor Rgl2-mediated nucleotide exchange of Ral GTPase. A high-resolution 1.18-Å X-ray cocrystal structure shows that the compound binds to a well-defined binding site in RalA as a result of a switch II loop conformational change. The structure, along with additional high-resolution crystal structures of several analogs in complex with RalA, confirm the importance of key hydrogen bond anchors between compound sulfone oxygen atoms and Ral backbone nitrogen atoms. Our discovery of a pocket with features found on known druggable sites and covalent modification of a bystander tyrosine residue present in Ral and Ras GTPases provide a strategy that could lead to therapeutic agent targeting oncogenic Ras mutants that are devoid of a cysteine nucleophile.
Tuesday, March 17, 2020
CM93, a novel covalent small molecule inhibitor targeting lung cancer with mutant EGFR
Qiwei Wang, Jing Ni, Tao Jiang, Hwan Geun Choi, Tinghu Zhang, Nathanael Gray, Jean J. Zhao
BioRXiv, 2020
doi: https://doi.org/10.1101/2020.03.09.984500
Epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs) have provided successful targeted therapies for patients with EGFR-mutant non-small-cell lung cancer (NSCLC). Osimertinib (AZD9291) is a third-generation irreversible EGFR TKI that has received regulatory approval for overcoming resistance mediated by the EGFR T790M mutation as well as a first-line treatment targeting EGFR activating mutations. However, a significant fraction of patients cannot tolerate the adverse effect associated with AZD9291. In addition, brain metastases are common in patients with NSCLN and remain a major clinical challenge. Here, we report the development of a novel third-generation EGFR TKI, CM93. Compared to AZD9291, CM93 exhibits improved lung cancer targeting and brain penetration and has demonstrated promising antitumor efficacy in mouse models of both EGFR-mutant NSCLC orthotopic and brain metastases. In addition, we find that CM93 confers superior safety benefits in mice. Our results demonstrate that further evaluations of CM93 in clinical studies for patients with EGFR-mutant NSCLC and brain metastases are warranted.
BioRXiv, 2020
doi: https://doi.org/10.1101/2020.03.09.984500
Epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs) have provided successful targeted therapies for patients with EGFR-mutant non-small-cell lung cancer (NSCLC). Osimertinib (AZD9291) is a third-generation irreversible EGFR TKI that has received regulatory approval for overcoming resistance mediated by the EGFR T790M mutation as well as a first-line treatment targeting EGFR activating mutations. However, a significant fraction of patients cannot tolerate the adverse effect associated with AZD9291. In addition, brain metastases are common in patients with NSCLN and remain a major clinical challenge. Here, we report the development of a novel third-generation EGFR TKI, CM93. Compared to AZD9291, CM93 exhibits improved lung cancer targeting and brain penetration and has demonstrated promising antitumor efficacy in mouse models of both EGFR-mutant NSCLC orthotopic and brain metastases. In addition, we find that CM93 confers superior safety benefits in mice. Our results demonstrate that further evaluations of CM93 in clinical studies for patients with EGFR-mutant NSCLC and brain metastases are warranted.
Friday, March 6, 2020
Discovery of Lysine-Targeted eIF4E Inhibitors through Covalent Docking
Xiaobo Wan, Tangpo Yang, Adolfo Cuesta, Xiaming Pang, Trent E. Balius, John J. Irwin, Brian K. Shoichet, and Jack Taunton
J. Am. Chem. Soc. 2020
doi: 10.1021/jacs.9b10377
Eukaryotic translation initiation factor 4E (eIF4E) binds the m7GTP cap structure at the 5′-end of mRNAs, stimulating the translation of proteins implicated in cancer cell growth and metastasis. eIF4E is a notoriously challenging target, and most of the reported inhibitors are negatively charged guanine analogues with negligible cell permeability. To overcome these challenges, we envisioned a covalent targeting strategy. As there are no cysteines near the eIF4E cap binding site, we developed a covalent docking approach focused on lysine. Taking advantage of a “make-on-demand” virtual library, we used covalent docking to identify arylsulfonyl fluorides that target a noncatalytic lysine (Lys162) in eIF4E. Guided by cocrystal structures, we elaborated arylsulfonyl fluoride 2 to 12, which to our knowledge is the first covalent eIF4E inhibitor with cellular activity. In addition to providing a new tool for acutely inactivating eIF4E in cells, our computational approach may offer a general strategy for developing selective lysine-targeted covalent ligands.
J. Am. Chem. Soc. 2020
doi: 10.1021/jacs.9b10377
Eukaryotic translation initiation factor 4E (eIF4E) binds the m7GTP cap structure at the 5′-end of mRNAs, stimulating the translation of proteins implicated in cancer cell growth and metastasis. eIF4E is a notoriously challenging target, and most of the reported inhibitors are negatively charged guanine analogues with negligible cell permeability. To overcome these challenges, we envisioned a covalent targeting strategy. As there are no cysteines near the eIF4E cap binding site, we developed a covalent docking approach focused on lysine. Taking advantage of a “make-on-demand” virtual library, we used covalent docking to identify arylsulfonyl fluorides that target a noncatalytic lysine (Lys162) in eIF4E. Guided by cocrystal structures, we elaborated arylsulfonyl fluoride 2 to 12, which to our knowledge is the first covalent eIF4E inhibitor with cellular activity. In addition to providing a new tool for acutely inactivating eIF4E in cells, our computational approach may offer a general strategy for developing selective lysine-targeted covalent ligands.
Wednesday, March 4, 2020
Targeting the PI5P4K Lipid Kinase Family in Cancer Using Covalent Inhibitors
Sindhu Carmen Sivakumaren, Hyeseok Shim, Tinghu Zhang, Fleur M. Ferguson, Mark R. Lundquist,
Christopher M. Browne, Hyuk-Soo Seo, Marcia N. Paddock, Theresa D. Manz, Baishan Jiang, Ming-Feng Hao, Pranav Krishnan, Diana G. Wang, T. Jonathan Yang, Nicholas P. Kwiatkowski, Scott B. Ficarro, James M. Cunningham, Jarrod A. Marto, Sirano Dhe-Paganon, Lewis C. Cantley, Nathanael S. Gray
Cell Chemical Biology, 2020
The PI5P4Ks have been demonstrated to be important for cancer cell proliferation and other diseases. However, the therapeutic potential of targeting these kinases is understudied due to a lack of potent, specific small molecules available. Here, we present the discovery and characterization of a pan-PI5P4K inhibitor, THZ-P1-2, that covalently targets cysteines on a disordered loop in PI5P4Kα/β/γ. THZ-P1-2 demonstrates cellular on-target engagement with limited off-targets across the kinome. AML/ALL cell lines were sensitive to THZ-P1-2, consistent with PI5P4K's reported role in leukemogenesis. THZ-P1-2 causes autophagosome clearance defects and upregulation in TFEB nuclear localization and target genes, disrupting autophagy in a covalent-dependent manner and phenocopying the effects of PI5P4K genetic deletion. Our studies demonstrate that PI5P4Ks are tractable targets, with THZ-P1-2 as a useful tool to further interrogate the therapeutic potential of PI5P4K inhibition and inform drug discovery campaigns for these lipid kinases in cancer metabolism and other autophagy-dependent disorders.
Covalent Small Molecules as Enabling Platforms for Drug Discovery
Dalton, S.E. and Campos, S.
ChemBioChem, 2020
doi: 10.1002/cbic.201900674
Covalent drugs have experienced significant renewed interest in drug discovery. This resurgence has been accompanied by a better understanding of the reactivity relationships required to engage selective covalent bonds between nucleophilic proteins and electrophilic small molecules. As a result, researchers have come to the realisation that covalent molecules could also represent useful and novel tools aimed at supporting medicinal chemistry programmes. This review surveys the increasing number of drug discovery platforms employing covalent chemistries, and highlights the utility of these techniques for identifying and characterising small molecules and biological targets.
ChemBioChem, 2020
doi: 10.1002/cbic.201900674
Covalent drugs have experienced significant renewed interest in drug discovery. This resurgence has been accompanied by a better understanding of the reactivity relationships required to engage selective covalent bonds between nucleophilic proteins and electrophilic small molecules. As a result, researchers have come to the realisation that covalent molecules could also represent useful and novel tools aimed at supporting medicinal chemistry programmes. This review surveys the increasing number of drug discovery platforms employing covalent chemistries, and highlights the utility of these techniques for identifying and characterising small molecules and biological targets.
Monday, March 2, 2020
Systematic identification of engineered methionines and oxaziridines for efficient, stable, and site-specific antibody bioconjugation [@Toste_Group, @christhechang
Susanna K. Elledge, Hai L. Tran, Alec H. Christian, Veronica Steri, Byron Hann, F. Dean Toste, Christopher J. Chang, James A. Wells
Proceedings of the National Academy of Sciences 2020
DOI: 10.1073/pnas.1920561117
Site-specific chemical modification of proteins remains a critical need for bioconjugation. Here we explore the key parameters needed for efficient, selective, and stable modification of methionine using recently developed oxaziridine reagents, called ReACT. We systematically tested various oxaziridine compounds, and scanned accessible and buried sites in a therapeutic antibody for breast cancer to determine the chemical and structural parameters for most stable and efficient modification. We show these adducts are highly stable over days and can support the delivery of toxic payloads to regress tumors in animals. These studies on this important chemical modification expand our capability to site-specifically modify proteins and antibodies for many applications.
Proceedings of the National Academy of Sciences 2020
DOI: 10.1073/pnas.1920561117
Site-specific chemical modification of proteins remains a critical need for bioconjugation. Here we explore the key parameters needed for efficient, selective, and stable modification of methionine using recently developed oxaziridine reagents, called ReACT. We systematically tested various oxaziridine compounds, and scanned accessible and buried sites in a therapeutic antibody for breast cancer to determine the chemical and structural parameters for most stable and efficient modification. We show these adducts are highly stable over days and can support the delivery of toxic payloads to regress tumors in animals. These studies on this important chemical modification expand our capability to site-specifically modify proteins and antibodies for many applications.
Cysteine-specific protein multi-functionalization and disulfide bridging using 3-bromo-5-methylene pyrrolones
Yingqian Zhang, Chuanlong Zang, Guoce An, Mengdi Shang, Zenghui Cui, Gong Chen, Zhen Xi & Chuanzheng Zhou
Nature Communications 2020
DOI: 10.1038/s41467-020-14757-4
Many reagents have been developed for cysteine-specific protein modification. However, few of them allow for multi-functionalization of a single Cys residue and disulfide bridging bioconjugation. Herein, we report 3-bromo-5-methylene pyrrolones (3Br-5MPs) as a simple, robust, and versatile class of reagents for cysteine-specific protein modification. These compounds can be facilely synthesized via a one-pot mild reaction and they show comparable tagging efficiency but higher cysteine specificity than the maleimide counterparts. The addition of cysteine to 3Br-5MPs generates conjugates that are amenable to secondary addition by another thiol or cysteine, making 3Br-5MPs valuable for multi-functionalization of a single cysteine and disulfide bridging bioconjugation. The labeling reaction and subsequent treatments are mild enough to produce stable and active protein conjugates for biological applications.
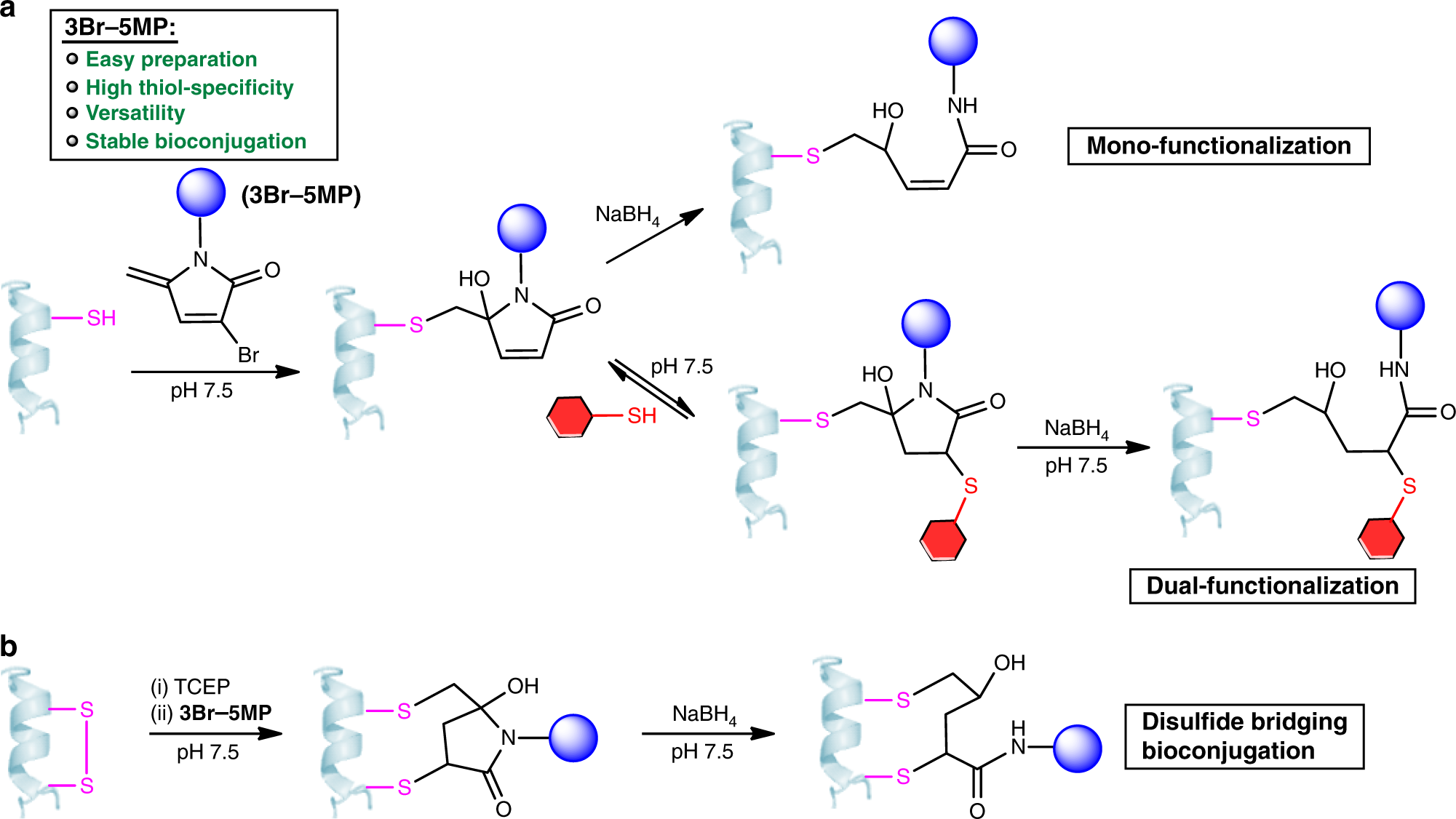
Nature Communications 2020
DOI: 10.1038/s41467-020-14757-4
Many reagents have been developed for cysteine-specific protein modification. However, few of them allow for multi-functionalization of a single Cys residue and disulfide bridging bioconjugation. Herein, we report 3-bromo-5-methylene pyrrolones (3Br-5MPs) as a simple, robust, and versatile class of reagents for cysteine-specific protein modification. These compounds can be facilely synthesized via a one-pot mild reaction and they show comparable tagging efficiency but higher cysteine specificity than the maleimide counterparts. The addition of cysteine to 3Br-5MPs generates conjugates that are amenable to secondary addition by another thiol or cysteine, making 3Br-5MPs valuable for multi-functionalization of a single cysteine and disulfide bridging bioconjugation. The labeling reaction and subsequent treatments are mild enough to produce stable and active protein conjugates for biological applications.

Re-evaluating the mechanism of action of α,β-unsaturated carbonyl DUB inhibitors b-AP15 and VLX1570: a paradigmatic example of unspecific protein crosslinking with Michael acceptor motif-containing drugs.
Jennifer A. Ward, Adan Pinto-Fernandez, Loic Cornelissen, Sarah Bonham, Laura Díaz-Sáez, Olivier Riant, Kilian V. M. Huber, Benedikt M Kessler, Olivier Feron, and Edward W. Tate
Journal of Medicinal Chemistry 2020
DOI: 10.1021/acs.jmedchem.0c00144
Deubiquitinating enzymes are a growing target class across multiple disease states, with several inhibitors now report-ed. b-AP15 and VLX1570 are two structurally related USP14/UCH-37 inhibitors. Through a proteomic approach, we demonstrate that these compounds target a diverse range of proteins, resulting in the formation of higher molecular weight complexes. Activity-based proteome profiling identified CIAPIN1 as a sub-micromolar covalent target of VLX1570, and further analysis demonstrated that high molecular weight complex formation leads to aggregation of CIAPIN1 in intact cells. Our results suggest that in addition to DUB inhibition, these compounds induce non-specific protein aggregation, providing a molecular explanation for general cellular toxicity.
Journal of Medicinal Chemistry 2020
DOI: 10.1021/acs.jmedchem.0c00144
Deubiquitinating enzymes are a growing target class across multiple disease states, with several inhibitors now report-ed. b-AP15 and VLX1570 are two structurally related USP14/UCH-37 inhibitors. Through a proteomic approach, we demonstrate that these compounds target a diverse range of proteins, resulting in the formation of higher molecular weight complexes. Activity-based proteome profiling identified CIAPIN1 as a sub-micromolar covalent target of VLX1570, and further analysis demonstrated that high molecular weight complex formation leads to aggregation of CIAPIN1 in intact cells. Our results suggest that in addition to DUB inhibition, these compounds induce non-specific protein aggregation, providing a molecular explanation for general cellular toxicity.
Subscribe to:
Posts (Atom)
Covalent Inhibitors of Monoacylglycerol Lipase Induce Conformational Changes and Proteasomal Degradation
Jordan A. Pham, Thanawat Thaingtamtanha, William McLeish, David Lefebvre, Spencer M. Uguccioni, Roxana Filip, Francesco Gentile, and John Pa...
-
Design, synthesis and biological evaluation of the activity-based probes for FGFR covalent inhibitorDandan Zhu, Zijian Zheng, Huixin Huang, Xiaojuan Chen, Shuhong Zhang, Zhuchu Chen, Ting Liu, Guangyu Xu, Ying Fu, Yongheng Chen, European Jo...
-
DOI Ansgar Oberheide, Maxime van den Oetelaar, Jakob Scheele, Jan Borggräfe, Semmy Engelen, Michael Sattler, Christian Ottmann, ...
-
Yoav Shamir, Nir London bioRxiv 2025.03.19.642201 doi: https://doi.org/10.1101/2025.03.19.642201 Recent years have seen an explosion in the...









