Jude Canon, Karen Rex, Anne Y. Saiki, Christopher Mohr, Keegan Cooke, Dhanashri Bagal,
Kevin Gaida, Tyler Holt, Charles G. Knutson, Neelima Koppada, Brian A. Lanman, Jonathan
Werner, Aaron S. Rapaport, Tisha San Miguel, Roberto Ortiz, Tao Osgood, Ji-Rong Sun,
Xiaochun Zhu, John D. McCarter, Laurie P. Volak, Brett E. Houk, Marwan G. Fakih, Bert H.
O’Neil, Timothy J. Price, Gerald S. Falchook, Jayesh Desai, James Kuo, Ramaswamy Govindan, David S. Hong, Wenjun Ouyang, Haby Henary, Tara Arvedson, Victor J. Cee & J. Russell Lipford
Nature 575, 217–223 (2019).
DOI: https://doi.org/10.1038/s41586-019-1694-1
KRAS is the most frequently mutated oncogene in cancer and encodes a key signalling protein in tumours. The KRAS(G12C) mutant has a cysteine residue that has been exploited to design covalent inhibitors that have promising preclinical activity. Here we optimized a series of inhibitors, using novel binding interactions to markedly enhance their potency and selectivity. Our efforts have led to the discovery of AMG 510, which is, to our knowledge, the first KRAS(G12C) inhibitor in clinical development. In preclinical analyses, treatment with AMG 510 led to the regression of KRASG12C tumours and improved the anti-tumour efficacy of chemotherapy and targeted agents. In immune-competent mice, treatment with AMG 510 resulted in a pro-inflammatory tumour microenvironment and produced durable cures alone as well as in combination with immune-checkpoint inhibitors. Cured mice rejected the growth of isogenic KRASG12D tumours, which suggests adaptive immunity against shared antigens. Furthermore, in clinical trials, AMG 510 demonstrated anti-tumour activity in the first dosing cohorts and represents a potentially transformative therapy for patients for whom effective treatments are lacking.
A blog highlighting recent publications in the area of covalent modification of proteins, particularly relating to covalent-modifier drugs. @CovalentMod on Twitter, @covalentmod@mstdn.science on Mastodon, and @covalentmod.bsky.social on BlueSky
Saturday, January 25, 2020
Monday, January 20, 2020
Structure-based design of a potent and selective covalent inhibitor for SRC kinase that targets a p-Loop cysteine
Guangyan Du, Suman Rao, Deepak Gurbani,
Nathaniel J. Henning, Jie Jiang, Jianwei Che, Annan Yang, Scott B Ficarro,
Jarrod A. Marto, Andrew J. Aguirre, Peter K. Sorger, Kenneth Dale Westover,
Tinghu Zhang, and Nathanael S Gray.
J. Med. Chem. 2020.DOI: https://doi.org/10.1021/acs.jmedchem.9b01502
SRC is a major regulator of many
signaling pathways and contributes to cancer development. However, development
of a selective SRC inhibitor has been challenging, and FDA-approved SRC
inhibitors, dasatinib and bosutinib, are multitargeted kinase inhibitors. Here,
we describe our efforts to develop a selective SRC covalent inhibitor by
targeting cysteine 277 on the P loop of SRC. Using a promiscuous covalent
kinase inhibitor (CKI) SM1-71 as a starting point we developed covalent
inhibitor 15a, which discriminates SRC from other covalent targets of SM1-71
including TAK1 and FGFR1. As an irreversible covalent inhibitor, compound 15a
exhibited sustained inhibition of SRC signaling both in vitro and in vivo.
Moreover, 15a exhibited potent anti-proliferative effects in non-small cell
lung cancer cell lines harboring SRC activation, thus providing evidence that
this approach may be promising for further drug development efforts.
Saturday, January 11, 2020
Structure-based design and analysis of SuFEx chemical probes
Lyn H. Jones and Jeffery W. Kelly
RSC Med. Chem., 2020
DOI: 10.1039/C9MD00542K
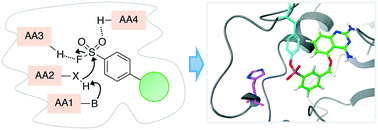
RSC Med. Chem., 2020
DOI: 10.1039/C9MD00542K
The discerning reactivity of sulfur(VI)-fluoride exchange (SuFEx) chemistry has enabled the context-specific labeling of protein binding sites by chemical probes that incorporate these versatile warheads. Emerging information from protein-probe structures and proteomic mapping experiments is helping advance our understanding of the protein microenvironment that dictates the reactivity of targetable amino acid residues. This review explores these new findings that should influence the future rational design of SuFEx probes for a multitude of applications in chemical biology and drug discovery.

Wednesday, January 8, 2020
Efficient targeted degradation via reversible and irreversible covalent PROTACs
Ronen Gabizon Amit Shraga Paul Gehrtz Ella Livnah Neta Gurwicz Liat Avram Tamar Unger Shira Albeck Ziv Shulman Nir London
ChemRxiv, 2020
DOI: 10.26434/chemrxiv.11494398.v1
PROteolysis Targeting Chimeras (PROTACs) represent an exciting inhibitory modality with many advantages, including sub-stoichiometric degradation of targets. Their scope, though, is still limited to-date by the requirement for a sufficiently potent target binder. A solution that proved useful in tackling challenging targets is the use of electrophiles to allow irreversible binding to the target. However, such binding will negate the catalytic nature of PROTACs. Reversible covalent PROTACs offer the best of both worlds. They possess the potency and selectivity associated with the formation of the covalent bond, while being able to dissociate and regenerate once the protein target is degraded. Using Bruton’s tyrosine kinase (BTK) as a clinically relevant model system, we present a proof-of concept for the first in class cyanoacrylamide reversible covalent PROTACs. We show efficient degradation with reversible covalent PROTACs, as well as their non-covalent and irreversible counterparts. The latter are amongst the most efficient PROTACs reported for BTK. They display single digit nM DC50, full degradation within 2-4 hours, proteome wide selectivity and show ~10-fold better inhibition of B cell activation than Ibrutinib. These examples refute the notion that covalent binders are not suitable as the basis for PROTACs, and may pave the way for the design of covalent PROTACs for a wide variety of challenging targets.
ChemRxiv, 2020
DOI: 10.26434/chemrxiv.11494398.v1
PROteolysis Targeting Chimeras (PROTACs) represent an exciting inhibitory modality with many advantages, including sub-stoichiometric degradation of targets. Their scope, though, is still limited to-date by the requirement for a sufficiently potent target binder. A solution that proved useful in tackling challenging targets is the use of electrophiles to allow irreversible binding to the target. However, such binding will negate the catalytic nature of PROTACs. Reversible covalent PROTACs offer the best of both worlds. They possess the potency and selectivity associated with the formation of the covalent bond, while being able to dissociate and regenerate once the protein target is degraded. Using Bruton’s tyrosine kinase (BTK) as a clinically relevant model system, we present a proof-of concept for the first in class cyanoacrylamide reversible covalent PROTACs. We show efficient degradation with reversible covalent PROTACs, as well as their non-covalent and irreversible counterparts. The latter are amongst the most efficient PROTACs reported for BTK. They display single digit nM DC50, full degradation within 2-4 hours, proteome wide selectivity and show ~10-fold better inhibition of B cell activation than Ibrutinib. These examples refute the notion that covalent binders are not suitable as the basis for PROTACs, and may pave the way for the design of covalent PROTACs for a wide variety of challenging targets.
Sunday, January 5, 2020
Tunable heteroaromatic sulfones enhance in-cell cysteine profiling
Hashim F Motiwala, Yu-Hsuan Kuo, Brittany L. Stinger, Bruce A. Palfey, and Brent R. Martin
Journal of the American Chemical Society 2020
DOI: 10.1021/jacs.9b08831Heteroaromatic sulfones react with cysteine via nucleophilic aromatic substitution, providing a mechanistically selective and irreversible scaffold for cysteine conjugation. Here we evaluate a library of heteroaromatic sulfides with different oxidation states, heteroatom substitutions, and a series of electron donating and electron-withdrawing substituents. Select substitutions profoundly influence reactivity and stability compared to conventional cysteine conjugation reagents, increasing the reaction rate by >3-orders of magnitude. The findings establish a series of synthetically accessible electrophilic scaffolds tunable across multiple tunable centers. New electrophiles and their corresponding alkyne-conjugates were profiled directly in cultured cells, achieving thiol saturation in a few minutes at sub-millimolar concentrations. Direct addition of desthiobiotin-functionalized probes to cultured cells simplified enrichment and elution to enable mass spectrometry discovery of >3000 reactive and/or accessible thiols labeled in their native cellular environments in a fraction of the standard analysis time. Surprisingly, only 1/2 of annotated cysteines were identified by both iodoacetamide-desthiobiotin and methylsulfonylbenzothiazole-desthiobiotin in replicate experiments, demonstrating complementary detection by mass spectrometry analysis. These probes offer advantages over existing cysteine alkylation reagents, including accelerated reaction rates, improved stability, and robust ionization for mass spectrometry applications. Overall, heteroaromatic sulfones provide modular tunability, shifted chromatographic elution times, and superior in-cell cysteine profiling for in-depth proteome-wide analysis and covalent ligand discovery.
Friday, January 3, 2020
Enhancing Intracellular Concentration and Target Engagement of PROTACs with Reversible Covalent Chemistry
Wen-Hao Guo, Xiaoli Qi, Yang Liu, Chan-I Chung, Fang Bai, Xingcheng Lin, Lingfei Wang, Jianwei Chen, Krystle J. Nomie, Feng Li, Meng C. Wang, Xiaokun Shu, José N. Onuchic, Jennifer A. Woyach, Michael L. Wang, Jin Wang
bioRxiv 2019
Current efforts in the proteolysis targeting chimera (PROTAC) field mostly focus on choosing the appropriate E3 ligase for a certain targeted protein, improving the binding affinities towards the target protein and the E3 ligase, and optimizing the PROTAC linker. However, it is well known that due to the large sizes of PROTAC molecules, their cellular uptake level remains an issue, posing a challenge to translate PROTACs into therapeutics. Driven by our fundamental investigation to compare how different warhead chemistry, reversible noncovalent (RNC), reversible covalent (RC), and irreversible covalent (IRC) binders, may affect the degradation of a model protein Bruton’s Tyrosine Kinase (BTK), we serendipitously discovered that cyano-acrylamide-based reversible covalent chemistry can significantly enhance the intracellular concentration and target engagement of the PROTAC. Building on this discovery, we developed RC-1 as the first reversible covalent BTK PROTAC, which has high target occupancy and is effective as both an inhibitor and a degrader. Molecular dynamics calculations and phase-separation based ternary complex assays support that RC-1 forms a stable ternary complex with BTK and Cereblon (CRBN). Additionally, RC-1 compares favorably with other reported BTK degraders in cell viability and target engagement assays and has a reasonable plasma half-life for in vivo applications. Importantly, this reversible covalent strategy can be generalized and applied to improve other PROTACs. This work can not only help to develop optimal BTK degraders for clinical applications but also provide a new strategy to improve PROTAC efficacy.
Thursday, January 2, 2020
Catalytic Mechanism and Covalent Inhibition of UDP-N-Acetylglucosamine Enolpyruvyl Transferase (MurA): Implications to the Design of Novel Antibacterials
Levente M. Mihalovits, György G. Ferenczy, and György M. Keserű
Journal of Chemical Information and Modeling 2019 59 (12), 5161-5173
DOI: 10.1021/acs.jcim.9b00691UDP-N-acetylglucosamine enolpyruvyl transferase (MurA) catalyzes the first step in the biosynthesis of the bacterial cell wall. This pathway is essential for the growth of bacteria but missing in mammals, that nominates MurA as an attractive antibacterial target. MurA has a flexible loop whose conformational change is known to be part of the activation mechanism of the enzyme. We have shown that the loop closed conformation makes the proton transfer from Cys115 to His394 possible by a low barrier exothermic process. QM/MM MD simulations revealed that the activated thiolate is able to react with phosphoenolpyruvate (PEP), the natural substrate of MurA. The binding free energy profile of several covalent inhibitors with various warheads reacting with the activated Cys115 was calculated by QM/MM MD simulations and confirmed that reaction barrier heights tend to separate active from inactive compounds. Our results give new insight into the catalytic mechanism and covalent inhibition of MurA and suggest that QM/MM MD simulations are able to support ligand design by providing sensible relative free energy barriers for covalent inhibitors with various warheads reacting with thiolate nucleophiles.
Subscribe to:
Posts (Atom)
Characterization of the Second-Generation Covalent Fragment Library (CovLib Gen2): Thiol Reactivity Profiling and p53-Y220C Rescue
Schwer, M., Aldea, S. R., Engelhardt, M. U., Stahlecker, J., Rheinganz, J., Langkamp, A., & Boeckler, F. M. Drug Design, Development an...
-
Design, synthesis and biological evaluation of the activity-based probes for FGFR covalent inhibitorDandan Zhu, Zijian Zheng, Huixin Huang, Xiaojuan Chen, Shuhong Zhang, Zhuchu Chen, Ting Liu, Guangyu Xu, Ying Fu, Yongheng Chen, European Jo...
-
DOI Ansgar Oberheide, Maxime van den Oetelaar, Jakob Scheele, Jan Borggräfe, Semmy Engelen, Michael Sattler, Christian Ottmann, ...
-
Yoav Shamir, Nir London bioRxiv 2025.03.19.642201 doi: https://doi.org/10.1101/2025.03.19.642201 Recent years have seen an explosion in the...




