Mélissanne de Wispelaere, Guangyan Du, Katherine A. Donovan, Tinghu Zhang, Nicholas A. Eleuteri, Jingting C. Yuan, Joann Kalabathula, Radosław P. Nowak, Eric S. Fischer, Nathanael S. Gray & Priscilla L. Yang
Nature Communications, 2019, 10, 3468
doi: 10.1038/s41467-019-11429-w
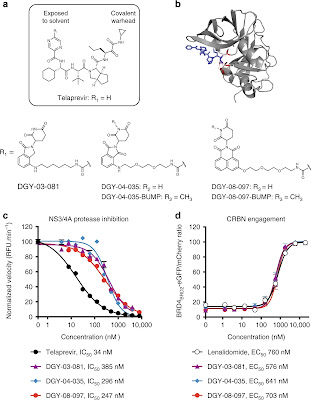
Targeted protein degradation is a promising drug development paradigm. Here we leverage this strategy to develop a new class of small molecule antivirals that induce proteasomal degradation of viral proteins. Telaprevir, a reversible-covalent inhibitor that binds to the hepatitis C virus (HCV) protease active site is conjugated to ligands that recruit the CRL4CRBN ligase complex, yielding compounds that can both inhibit and induce the degradation of the HCV NS3/4A protease. An optimized degrader, DGY-08-097, potently inhibits HCV in a cellular infection model, and we demonstrate that protein degradation contributes to its antiviral activity. Finally, we show that this new class of antiviral agents can overcome viral variants that confer resistance to traditional enzymatic inhibitors such as telaprevir. Overall, our work provides proof-of-concept that targeted protein degradation may provide a new paradigm for the development of antivirals with superior resistance profiles.
A blog highlighting recent publications in the area of covalent modification of proteins, particularly relating to covalent-modifier drugs. @CovalentMod on Twitter, @covalentmod@mstdn.science on Mastodon, and @covalentmod.bsky.social on BlueSky
Linking of fragments in neighboring binding sites is one of the optimization strategies in fragment-based drug discovery, where additive or even more substantial bioactivity improvements can be realized. However, such efforts present a considerable challenge when one fragment binds covalently to the target protein, as small modifications can influence the correct positioning of the covalent warhead toward the targeted nucleophilic residue. Here, we present a case study of fragment linking that yielded single-digit micromolar, covalent inhibitors of the SARS-CoV-2 main protease, starting from fragments that were inactive in the biochemical assay. Using structural information from a recent, high-throughput crystallographic fragment screen, we show that the success of fragment linking in the design of targeted covalent inhibitors is heavily impacted by several factors, including the warhead type, the labeling chemistry, and even subtle changes in the designed linker. Notably, we observe that induced fit effects might override the original fragment orientations in the linked molecule, highlighting the need for reliable structure verification, especially in consecutive rounds of fragment elaboration.
Levente Kollár, Levente M. Mihalovits, Dávid Bajusz, DamijanKnez, József Simon, Blake H. Balcomb, Daren Fearon, Stanislav Gobec, György M. K...

-
Design, synthesis and biological evaluation of the activity-based probes for FGFR covalent inhibitorDandan Zhu, Zijian Zheng, Huixin Huang, Xiaojuan Chen, Shuhong Zhang, Zhuchu Chen, Ting Liu, Guangyu Xu, Ying Fu, Yongheng Chen, European Jo...
-
DOI Ansgar Oberheide, Maxime van den Oetelaar, Jakob Scheele, Jan Borggräfe, Semmy Engelen, Michael Sattler, Christian Ottmann, ...
-
Yoav Shamir, Nir London bioRxiv 2025.03.19.642201 doi: https://doi.org/10.1101/2025.03.19.642201 Recent years have seen an explosion in the...