Katharine E. Gilbert, Aini Vuorinen, Arron Aatkar, Peter Pogány, Jonathan Pettinger, Emma K. Grant, Joanna M. Kirkpatrick, Katrin Rittinger, David House, Glenn A. Burley, and Jacob T. Bush
A blog highlighting recent publications in the area of covalent modification of proteins, particularly relating to covalent-modifier drugs. @CovalentMod on Twitter, @covalentmod@mstdn.science on Mastodon, and @covalentmod.bsky.social on BlueSky
Saturday, January 21, 2023
Profiling Sulfur(VI) Fluorides as Reactive Functionalities for Chemical Biology Tools and Expansion of the Ligandable Proteome
Friday, January 20, 2023
Fast and Bioorthogonal Release of Isocyanates in Living Cells from Iminosydnones and Cycloalkynes
Maxime Ribéraud, Karine Porte, Arnaud Chevalier, Léa Madegard, Aurélie Rachet, Agnès Delaunay-Moisan, Florian Vinchon, Pierre Thuéry, Giovanni Chiappetta, Pier Alexandre Champagne, Grégory Pieters, Davide Audisio, and Frédéric Taran
Monday, January 9, 2023
Discovery of Potent Small-Molecule Inhibitors of WDR5-MYC Interaction
Jian Ding, Guo Li, Hejun Liu, Lulu Liu, Ying Lin, Jingyan Gao, Guoqiang Zhou, Lingling Shen, Mengxi Zhao, Yanyan Yu, Weihui Guo, Ulrich Hommel, Johannes Ottl, Jutta Blank, Nicola Aubin, Yi Wei, Hu He, David R. Sage, Peter W. Atadja, En Li, Rishi K. Jain, John A. Tallarico, Stephen M. Canham, Ying-Ling Chiang, and He Wang
ACS Chemical Biology 2023
DOI: 10.1021/acschembio.2c00843
Friday, January 6, 2023
Inhibiting androgen receptor splice variants with cysteine-selective irreversible covalent inhibitors to treat prostate cancer
Thirumagal Thiyagarajan, Suriyan Ponnusamy, Dong-Jin Hwang, Yali He, Sarah Asemota, Kirsten L Young, Daniel L Johnson, Vera Bocharova, Weidong Zhou, Abhinav K Jain, Emanuel F Petricoin 5, Zheng Yin, Lawrence M Pfeffer, Duane D Miller, Ramesh Narayanan
PNAS, 120 (1) e2211832120
https://doi.org/10.1073/pnas.2211832120
Androgen receptor (AR) and its splice variants (AR-SVs) promote prostate cancer (PCa) growth by orchestrating transcriptional reprogramming. Mechanisms by which the low complexity and intrinsically disordered primary transactivation domain (AF-1) of AR and AR-SVs regulate transcriptional programming in PCa remains poorly defined. Using omics, live and fixed fluorescent microscopy of cells, and purified AF-1 and AR-V7 recombinant proteins we show here that AF-1 and the AR-V7 splice variant form molecular condensates by liquid–liquid phase separation (LLPS) that exhibit disorder characteristics such as rapid intracellular mobility, coactivator interaction, and euchromatin induction. The LLPS and other disorder characteristics were reversed by a class of small-molecule-selective AR-irreversible covalent antagonists (SARICA) represented herein by UT-143 that covalently and selectively bind to C406 and C327 in the AF-1 region. Interfering with LLPS formation with UT-143 or mutagenesis resulted in chromatin condensation and dissociation of AR-V7 interactome, all culminating in a transcriptionally incompetent complex. Biochemical studies suggest that C327 and C406 in the AF-1 region are critical for condensate formation, AR-V7 function, and UT-143’s irreversible AR inhibition. Therapeutically, UT-143 possesses drug-like pharmacokinetics and metabolism properties and inhibits PCa cell proliferation and tumor growth. Our work provides critical information suggesting that clinically important AR-V7 forms transcriptionally competent molecular condensates and covalently engaging C327 and C406 in AF-1, dissolves the condensates, and inhibits its function. The work also identifies a library of AF-1-binding AR and AR-SV-selective covalent inhibitors for the treatment of PCa.
Novel Irreversible Peptidic Inhibitors of Transglutaminase 2
Nicholas J. Cundy, Jane Arciszewski, Eric W. J. Gates, Sydney L. Acton, Kyle D. Passley, Ernest Awoonor-Williams, Elizabeth K. Boyd, Nancy Xu, Elise Pierson, Catalina Fernandez-Ansieta, Marie R. Albert, Nicole M. R. McNeil, Gautam Adhikary, Richard L. Eckert and Jeffrey W Keillor
RSC Medicinal Chemistry, 2023
DOI https://doi.org/10.1039/D2MD00417H
Transglutaminase 2 (TG2), also referred to as tissue transglutaminase, is an enzyme that plays crucial roles in both protein crosslinking and cell signalling. It is capable of both catalysing transamidation and acting as a G-protein, these activities being conformation-dependent, mutually exclusive and tightly regulated. The dysregulation of both activities has been implicated in numerous pathologies. TG2 is expressed ubiquitously in humans and is localized both intracellularly and extracellularly. Targeted TG2 therapies have been developed but have faced numerous hurdles including decreased efficacy in vivo. Our latest efforts in inhibitor optimization involve the modification of a previous lead compound’s scaffold by insertion of various amino acid residues into the peptidomimetic backbone, and derivatization of the N-terminus with substituted phenylacetic acids, resulting in 28 novel irreversible inhibitors. These inhibitors were evaluated for their ability to inhibit TG2 in vitro and their pharmacokinetic properties, and the most promising candidate was tested in a cancer stem cell model. Although these inhibitors display exceptional potency versus TG2, with kinact/KI ratios nearly ten-fold higher than their parent compound, their pharmacokinetic properties and cellular activity limit their therapeutic potential. However, they do serve as a scaffold for the development of potent research tools.
Monday, January 2, 2023
β-Lactamases Evolve against Antibiotics by Acquiring Large Active-Site Electric Fields
Zhe Ji and Steven G. Boxer
Journal of the American Chemical Society 2022 144 (48), 22289-22294
DOI: 10.1021/jacs.2c10791
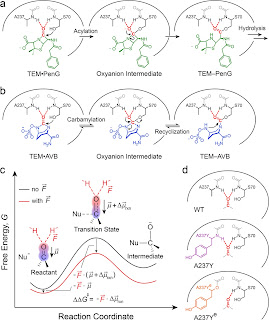
A compound bound covalently to an enzyme active site can act either as a substrate if the covalent linkage is readily broken up by the enzyme or as an inhibitor if the bond dissociates slowly. We tracked the reactivity of such bonds associated with the rise of the resistance to penicillin G (PenG) in protein evolution from penicillin-binding proteins (PBPs) to TEM β-lactamases and with the development of avibactam (Avb) to overcome the resistance. We found that the ester linkage in PBP–PenG is resistant to hydrolysis mainly due to the small electric fields present in the protein active site. Conversely, the same linkage in the descendant TEM–PenG experiences large electric fields that stabilize the more charge-separated transition state and thus lower the free energy barrier to hydrolysis. Specifically, the electric fields were improved from −59 to −140 MV/cm in an ancient evolution dating back billions of years, contributing 5 orders of magnitude rate acceleration. This trend continues today in the nullification of newly developed antibiotic drugs. The fast linkage hydrolysis acquired from evolution is counteracted by the upgrade of PenG to Avb whose linkage escapes from the hydrolysis by returning to a low-field environment. Using the framework of electrostatic catalysis, the electric field, an observable from vibrational spectroscopy, provides a unifying physical metric to understand protein evolution and to guide the design of covalent drugs.
Protein Electric Fields Enable Faster and Longer-Lasting Covalent Inhibition of β-Lactamases
Zhe Ji, Jacek Kozuch, Irimpan I. Mathews, Christian S. Diercks, Yasmin Shamsudin, Mirjam A. Schulz, and Steven G. Boxer
Journal of the American Chemical Society 2022 144 (45), 20947-20954
DOI: 10.1021/jacs.2c10791
The widespread design of covalent drugs has focused on crafting reactive groups of proper electrophilicity and positioning toward targeted amino-acid nucleophiles. We found that environmental electric fields projected onto a reactive chemical bond, an overlooked design element, play essential roles in the covalent inhibition of TEM-1 β-lactamase by avibactam. Using the vibrational Stark effect, the magnitudes of the electric fields that are exerted by TEM active sites onto avibactam’s reactive C═O were measured and demonstrate an electrostatic gating effect that promotes bond formation yet relatively suppresses the reverse dissociation. These results suggest new principles of covalent drug design and off-target site prediction. Unlike shape and electrostatic complementary which address binding constants, electrostatic catalysis drives reaction rates, essential for covalent inhibition, and deepens our understanding of chemical reactivity, selectivity, and stability in complex systems.
Covalent Ligand Electrophiles Are Differentially Activated by Proximity Effects Which Govern Latent Protein Reactivit
Tomas V. Frankovich, Harrison M. McCann, Kyle S. Hoffman, and Anthony F. Rullo ACS Central Science Article ASAP DOI: 10.1021/acscentsci....
-
DOI Ansgar Oberheide, Maxime van den Oetelaar, Jakob Scheele, Jan Borggräfe, Semmy Engelen, Michael Sattler, Christian Ottmann, ...
-
Design, synthesis and biological evaluation of the activity-based probes for FGFR covalent inhibitorDandan Zhu, Zijian Zheng, Huixin Huang, Xiaojuan Chen, Shuhong Zhang, Zhuchu Chen, Ting Liu, Guangyu Xu, Ying Fu, Yongheng Chen, European Jo...
-
Yoav Shamir, Nir London bioRxiv 2025.03.19.642201 doi: https://doi.org/10.1101/2025.03.19.642201 Recent years have seen an explosion in the...